I doubt that they would bind, but I never would have believed this possible in the first place.Īn absolutely brilliant piece of physical chemistry explained a puzzling biologic phenomenon that organic chemistry was powerless to illuminate. It would be amusing to do the same thing for a GPCR binding one of the monamines. So just solublizing the GPCR without changing any of the amino acids external to the membrane, produces an object for study. CCL5 is an 8 kiloDalton protein (probably 80 amino acids, while CXCL12 has 93. Why these two GPCR’s? Because their ligands are proteins and can’t snuggle deep down inside the 7 alpha helices embedded in the membrane (they’re just too big), but bind to the outside surface. You either have to remove them en bloc from the membrane or dissolve them in other lipids so they don’t denature. Why did they even do it? Because GPCR structures are hard to study. Instead of destroying the structure of the GPCRs (CCR5 and CXCR4) they became water soluble, and bound their ligands CCL5 for CCR5 and CXCL12 for CXCR4 to the same extent.Įven more amazing, the QTYdesigned receptors exhibit remarkable thermostability in the presence of arginine and retained ligand-binding activity after heat treatment at 60 ☌ for 4 h and 24 h, and at 100 ☌ for 10 min. Thus leucine (L - single amino acid letter code) is replaced by glutamine (Q), Isoleucine (I) and Valine (V) is replaced by Threonine (T) and finally phenylalanine (F) is replaced by Tyrosine (Y). The trick was to replace hydrophobic amino acids with hydrophilic ones with the same shape. So if you changed the hydrophobic amino acids found in the 7 transmembrane segments of GPCRs to hydrophilic ones - all hell should break loose.
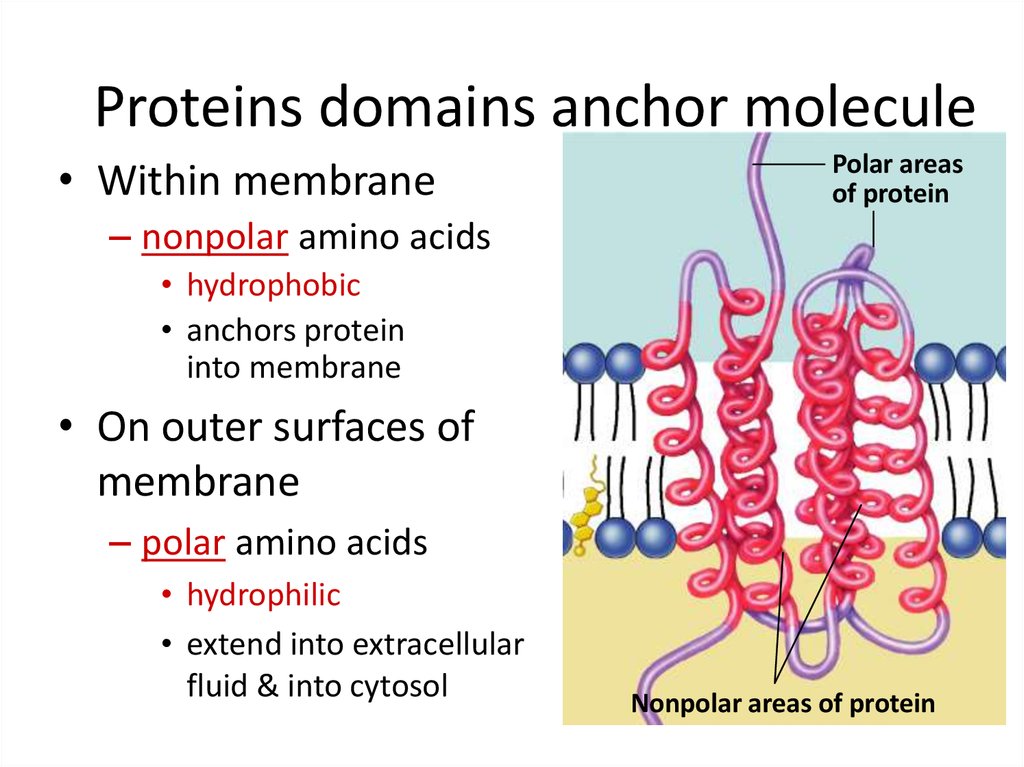
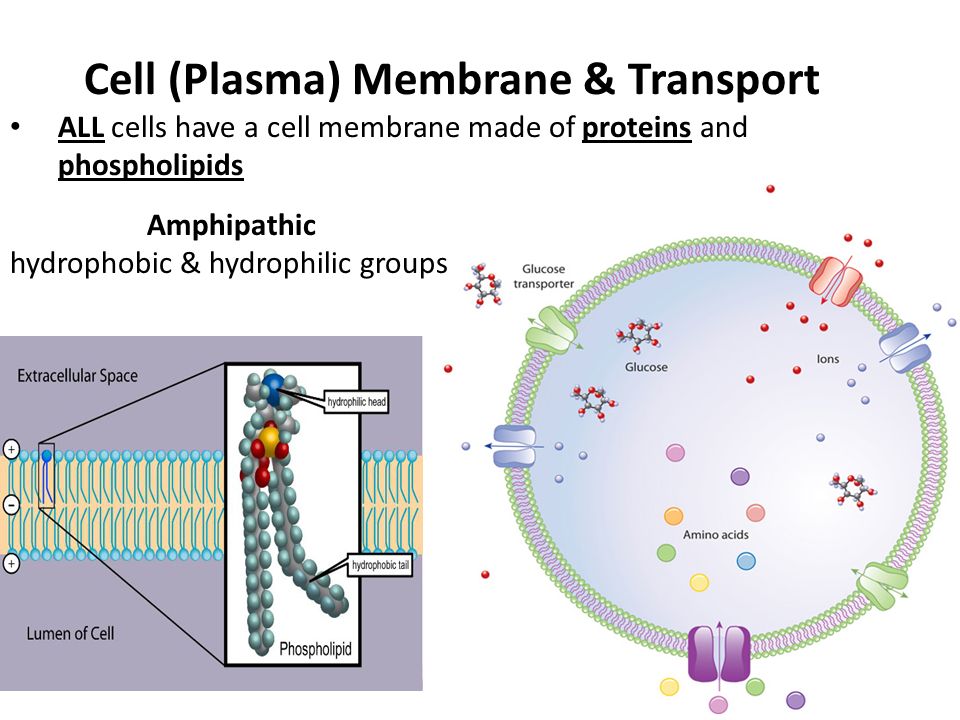
We estimate that ∼700 approved drugs target GPCRs, implying that approximately 35% of approved drugs target GPCRs.” GPCRs have been intensively by chemists, molecular biologists, pharmacologists and drug chemists with the net result that as of last year “128 GPCRs are targets for drugs listed in the Food and Drug Administration Orange Book. We ‘know’ that the 7 transmembrane alpha helices of G Protein Coupled Receptors (GPCRs) all contain hydrophobic amino acids, so they dissolve in the (hydrophobic) lipids of the membrane. How little we really understand about proteins.

